Thanks Jim, That is exactly the "toy" that I bought from BoatZincs.com. I am on a mooring most of the time. I'd like to see your setup for testing leads.Phosphate is being use in batteries now. LiFePO4 type. So they may act like the Cathode on a battery.
So the phosphates would draw Electrons from the Zinc, therefore making the Zinc sacrifice more than normal.
But they should not interfere with your testing.
I have this Probe...
Corrosion Reference Electrode: Corrosion Quiz
Jim...
Galvanic Corrosion
- Thread starter gfrazzle
- Start date
To Jim's procedure I would add record the boats that are around you when you test. If you get an anomalous reading it could be a neighboring boat causing the problem or if you get inconsistent readings it may be neighboring boats.Phosphate is being use in batteries now. LiFePO4 type. So they may act like the Cathode on a battery.
So the phosphates would draw Electrons from the Zinc, therefore making the Zinc sacrifice more than normal.
But they should not interfere with your testing.
I have this Probe...
Corrosion Reference Electrode: Corrosion Quiz
That comes with a proprietary manual on testing sequences. I called them and asked for a PDF file of their manual.
The owner said NOPE, since he is a certified ABYC electrician. So he is selling his knowledge too.
Here is what I suggest.
1) Record your testing volts and sea water temperature [I keep mine in a notebook for comparison ]
2) Drop the AgCl probe at the ≈ depth of your shaft Zinc and close to it.
3) Then test the shaft and wait till your meter stabilizes. [I have a alligator clip on a long testing wire to clamp on testing points] Next time on boat I will take a picture of my testing clamps.
4) If you have Zinc protection it should be close to the range on that chart above.
5) Disconnect shore power, see if the meter changes. No significant changes then you are not getting anything from the AC green wire.
Then proceeded to test any component on your boat, step by step.
One example noted that no problem until he turned on his radar. The radar transmission was not the problem but the radar supply circuit was bleeding.
I freaked out the first time I tested each of my bronze thru hull valves. I got -.470 V , [not right] until I realized it was my Lead-Antimony Keel I was reading.
My probe bought me a lot of...
Peace of Mind
Jim...
Here is a cool way to use your AgCl probe as a...If you get an anomalous reading it could be a neighboring boat causing the problem
Marina Stray DC current detector
The concept is like a Radio Frequency Direction finder.
1) Dangle and submerge the AgCl probe off one end of a 8' PVC pipe.
2) Dangle and submerge a 6" piece of Copper tubing on the opposite end of PVC pipe
[ connect the Copper to the other side of your meter ]
Now you have a "Spread Antenna field" between the AgCl and Copper.
3) Move "Detector" slowly in a small boat, around the Marina, to find the "Anomaly" in your meter reading.
_____
I suggest that a new boat in the marina needs to be "verified" using something like the Detector.
_____
Since my boat is 43' and I got tired of moving the Meter around, I built a long extension cord.
I started with an old flexible Xmas Light extension cord that when split gave me 80 feet long.
On the meter end I put one of these that fit my DVM...
Amazon.com: uxcell 4 Pcs Screw Type Audio Speaker Connector Banana Red Black: Electronics
On the other end for clamp testing , I put in two size clamps...
1) small alligator clamps like this
Amazon.com: KAIWEETS Electrical Alligator Clips Test Leads Sets Soldered and Stamping Jumper Wires for Circuit Connection/Experiment, 21 inches 5 Colors (10 PCS): Industrial & Scientific
2) Small battery clamps like the size on this.
Amazon.com: Electop Cigarette Lighter Battery Clamps, 12V 24V Battery Clip-on Car Cigarette Lighter Socket Adapter, 10Ft 16 AWG Cigarette Lighter Extension Cord with 10A Fuse: Electop
It took a while to build it but it can test everything and quickly.
You will need the clamps to reach awkward spaces and clamp on circuit wire.
Jim...
PS: I will send a picture of the set up soon.
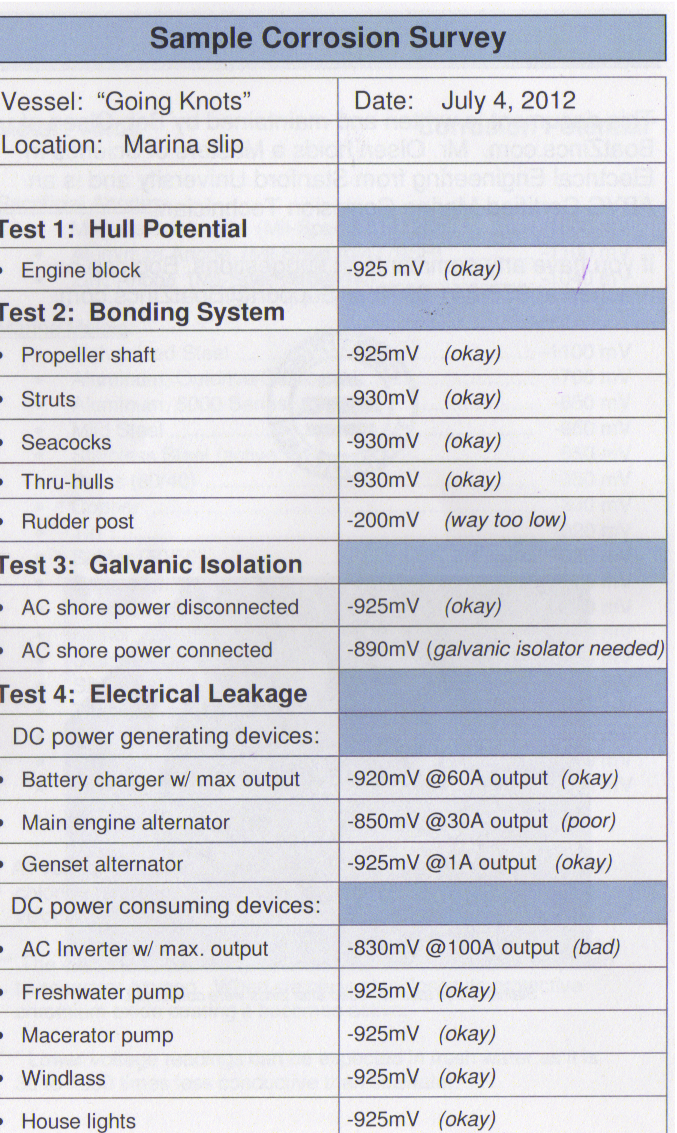
Here is a Sample Survey from the manual. I dont think, Mr Olsen, the ABYC corrosion expert would mind.
Jim...
Jim...
Attachments
-
1.8 MB Views: 138

